https://arcview-botanixpharma.s3.ap-southeast-2.amazonaws.com/botanixpharma.com/assets/img/2026/04/26-04-29-4c-Apr-Q-REPORTS-RELEASED-GRAPHIC-Final-.png
2026-04-29 09:24:00
2026-04-29 09:24:00
Botanix Releases Quarterly Reports
https://arcview-botanixpharma.s3.ap-southeast-2.amazonaws.com/botanixpharma.com/assets/img/2026/04/26-04-29-4c-Apr-Q-REPORTS-RELEASED-GRAPHIC-Final-.png
2026-04-29 09:24:00
2026-04-29 09:24:00
Botanix Releases Quarterly Reports
Latest news
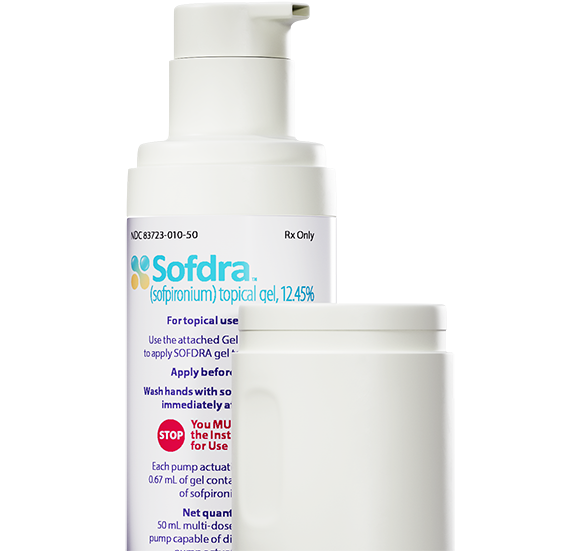
Sofdra ® (sofpironium) topical gel, 12.45% is now available in the US
Our lead dermatology product, Sofdra , is approved for use by the US Food and Drug Administration (FDA).
Innovators and commercial leaders in dermatology
Here at Botanix, we are at the forefront of dermatological innovation. With a world-class team and significant capital, we are committed to addressing common yet underserved skin diseases and infections.
As an ASX-listed company, we are in a prime position to develop and commercialise groundbreaking treatments across broad markets, all while adhering to the highest clinical standards set by the US FDA.
Check out our latest news